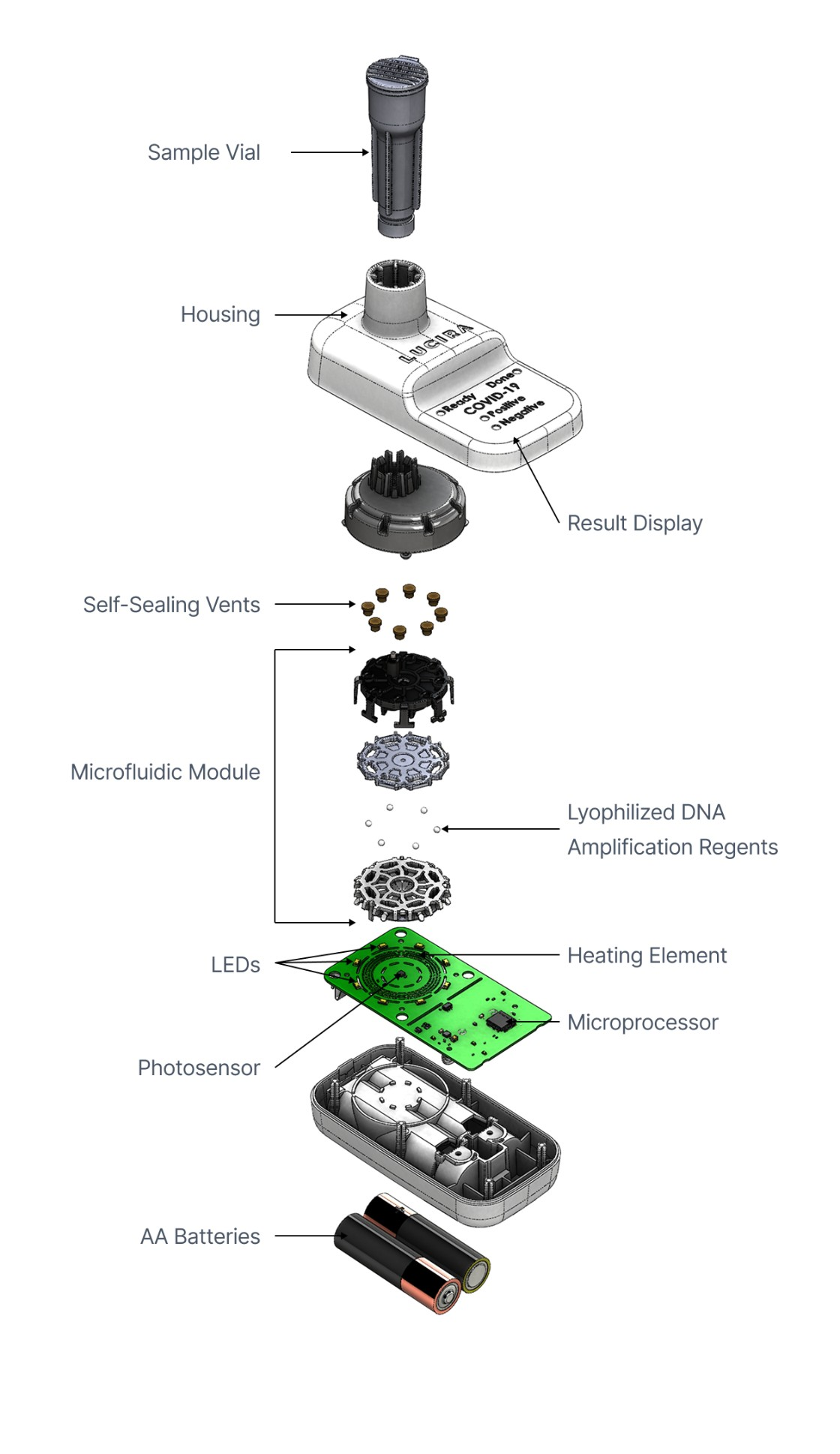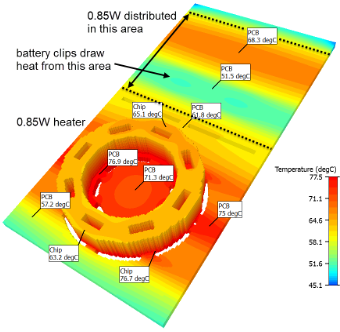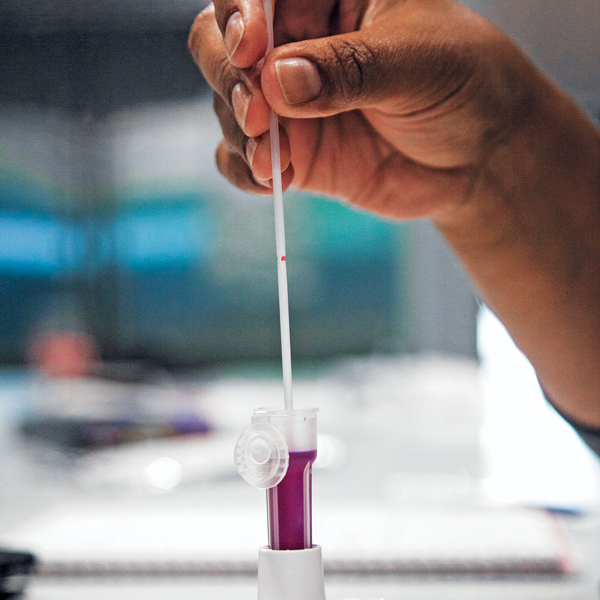
Lyophilized Reagents
Challenge
The product featured a polymerase enzyme and other assay components which needed to be lyophilized for room temperature stability.
Solution
We chose pellet lyophilization for its manufacturing scaling advantages, building an in-house dry room with robotic pick-and-place processes and working closely with lyophilization partners on pellet integrity and drying protocols.